Pseudomonas aeruginosa is classified as a Gram-negative bacteria. It can be described as an opportunistic microorganism since it possesses the ability to colonize bodies of vulnerable patients, especially critically ill ones or those suffering from cystic fibrosis. Pseudomonas aeruginosa is one of the major causes of Hospital-acquired infection worldwide and a represents a serious threat to Public Health. [1]
Pseudomonas aeruginosa can colonize many natural environments like soil, water and skin, because of its faculty to utilize a wide range of organic matter and cope with different environmental conditions. Pseudomonas aeruginosa colonizes human hosts, therefore it can use cysteine and methionine as sulfur a source of energy. [2]
Function
The thiol dioxygenation represents the initial oxidation step which allows the incorporation of a thiol to catabolic and biosynthetic pathways. A family of specific non-heme mononuclear iron proteins are necessary to catalyse this reaction. During the reaction, each enzyme reacts efficiently with only one substrate. This family of enzymes is made up of cysteine dioxygenase, cysteamine dioxygenase, mercaptosuccinate dioxygenase and 3-mercaptopropionate dioxygenase. [3]
The thiol dioxygenase of Pseudomonas aeruginosa is a 3-mercaptopropionate dioxygenase (p3MDO) with a secondary cysteine dioxygenase activity. Therefore, it can also be named 3-mercaptopropionate dioxygenase or cysteine dioxygenase.
This is the first example of cysteine dioxygenase homologue which utilizes a second substrate with near stochiometric coupling to dioxygen consumption.[4]
The cysteine dioxygenase homologue from Pseudomonas aeruginosa is expressed in low levels so this metabolic pathway is present in this organism.
The physiological role is unclear.
Interactions: Catalytic activity
Thiol dioxygenase of
Pseudomonas aeruginosa[5]
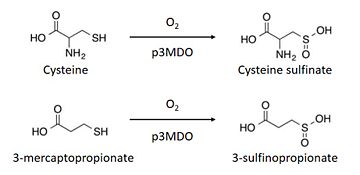
The thiol dioxygenase catalyses the dioxygenation of 3-mercaptopropionate to 3-sulfinopropionate. [6]
The substrate naturally binds itself to the ferrous iron through the thiol. However, spectroscopy indicates that each substrate can bind themselves to other parts of the enzyme as well. [7]
It also oxidizes cysteine to cysteine sulfinate. [8]
This enzyme has a marked preference for 3-mercaptopronionate, which explains why it is also referred to as a 3-mercaptopropionate dioxygenase.[9]
Structural highlights
Thiol dioxygenases share a common structure described as a 6-stranded β-barrel core, and a canonical cupin or “jelly roll” β-barrel that is formed with cupin motif 1, an intermotif region, and cupin motif 2 each forming two of the core six β-strands in the folded protein structure.[10]
The Thiol dioxygenase from Pseudomonas aeruginosa is made up of 4 chains (named , , , )[11]. Each chain is made up of 211 amino acids and has a molecular mass of 23 kDa. For its secondary structures, it has and [12]. Three Histidines, especially,, and constitute the binding sites of iron atom.
Furthermore, this protein has two different domains, namely a RmlC-like jelly roll fold and a RmlC-like cupin domain[13].
Disease
A mammalian cysteine dioxygenase also exists and its active site is slightly different, because of the presence of a glutamine instead of an arginine. [14]
In humans, patients with a high level of cysteine and glutathione-cysteine mixed with disulphide are most likely to suffer from Hallervorden-Spatz (HS) syndrome which is essentially characterised by neurochemical abnormalities since it affects the globus pallidus.
Patients suffering from this disease have been observed to have a low level in cysteine dioxygenase activity.
Thus, in this case, cysteine accumulates locally in the globus palladus and the conversion of cysteine to taurine is blocked due to enzymatic inactivity.
An increasing amount of cysteine may increase the level of iron content in Hallervorden-Spatz disease and the additive effects of an accumulated amount of cysteine and of ferrous iron may give rise to the formation of free radicals that can damage neuronal membranes to cause the typical morphological changes observed in this disorder. [15]

