Transmembrane protease serine 2
From Proteopedia
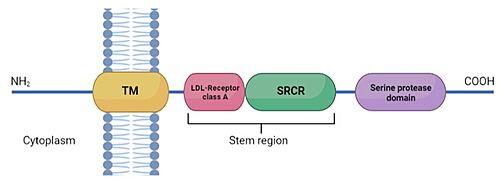
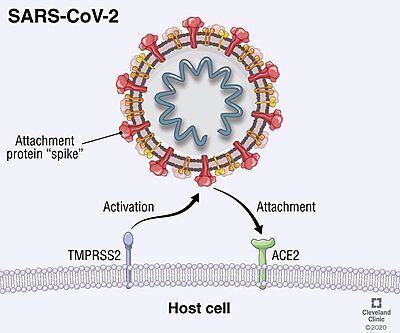
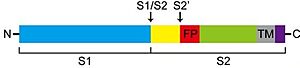
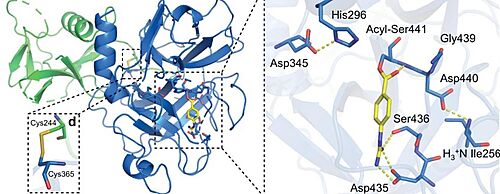
TMPRSS2 (Transmembrane protease serine 2) is a membrane protein belonging to the type II transmembrane serine protease (TTSP) family. It is functionally classified as a trypsin-like protease (TLP). [1] Serine proteases are known to be involved in many physiological and pathological processes, but the exact function remains unclear. It is involved in two main pathological processes, prostate cancer and viral entry. TMPRSS2 is expressed in several tissues regulated by androgens and undergoes autocatalytic cleavage for its activation.
| |||||||||||
References
- ↑ Sgrignani J, Cavalli A. Computational Identification of a Putative Allosteric Binding Pocket in TMPRSS2. Front Mol Biosci. 2021 Apr 30;8:666626. doi: 10.3389/fmolb.2021.666626., eCollection 2021. PMID:33996911 doi:http://dx.doi.org/10.3389/fmolb.2021.666626
- ↑ Evnin LB, Vasquez JR, Craik CS. Substrate specificity of trypsin investigated by using a genetic selection. Proc Natl Acad Sci U S A. 1990 Sep;87(17):6659-63. doi: 10.1073/pnas.87.17.6659. PMID:2204062 doi:http://dx.doi.org/10.1073/pnas.87.17.6659
- ↑ Singh N, Decroly E, Khatib AM, Villoutreix BO. Structure-based drug repositioning over the human TMPRSS2 protease domain: search for chemical probes able to repress SARS-CoV-2 Spike protein cleavages. Eur J Pharm Sci. 2020 Oct 1;153:105495. doi: 10.1016/j.ejps.2020.105495. Epub, 2020 Jul 28. PMID:32730844 doi:http://dx.doi.org/10.1016/j.ejps.2020.105495
- ↑ Thunders M, Delahunt B. Gene of the month: TMPRSS2 (transmembrane serine protease 2). J Clin Pathol. 2020 Dec;73(12):773-776. doi: 10.1136/jclinpath-2020-206987. Epub , 2020 Sep 1. PMID:32873700 doi:http://dx.doi.org/10.1136/jclinpath-2020-206987
- ↑ . PMID:216315890657
- ↑ Afar DE, Vivanco I, Hubert RS, Kuo J, Chen E, Saffran DC, Raitano AB, Jakobovits A. Catalytic cleavage of the androgen-regulated TMPRSS2 protease results in its secretion by prostate and prostate cancer epithelia. Cancer Res. 2001 Feb 15;61(4):1686-92. PMID:11245484
- ↑ Bertram S, Glowacka I, Blazejewska P, Soilleux E, Allen P, Danisch S, Steffen I, Choi SY, Park Y, Schneider H, Schughart K, Pohlmann S. TMPRSS2 and TMPRSS4 facilitate trypsin-independent spread of influenza virus in Caco-2 cells. J Virol. 2010 Oct;84(19):10016-25. doi: 10.1128/JVI.00239-10. Epub 2010 Jul 14. PMID:20631123 doi:http://dx.doi.org/10.1128/JVI.00239-10
- ↑ Shen LW, Mao HJ, Wu YL, Tanaka Y, Zhang W. TMPRSS2: A potential target for treatment of influenza virus and coronavirus infections. Biochimie. 2017 Nov;142:1-10. doi: 10.1016/j.biochi.2017.07.016. Epub 2017 Aug 1. PMID:28778717 doi:http://dx.doi.org/10.1016/j.biochi.2017.07.016
- ↑ Lam DK, Dang D, Flynn AN, Hardt M, Schmidt BL. TMPRSS2, a novel membrane-anchored mediator in cancer pain. Pain. 2015 May;156(5):923-930. doi: 10.1097/j.pain.0000000000000130. PMID:25734995 doi:http://dx.doi.org/10.1097/j.pain.0000000000000130
- ↑ Kim TS, Heinlein C, Hackman RC, Nelson PS. Phenotypic analysis of mice lacking the Tmprss2-encoded protease. Mol Cell Biol. 2006 Feb;26(3):965-75. doi: 10.1128/MCB.26.3.965-975.2006. PMID:16428450 doi:http://dx.doi.org/10.1128/MCB.26.3.965-975.2006
- ↑ St John J, Powell K, Conley-Lacomb MK, Chinni SR. TMPRSS2-ERG Fusion Gene Expression in Prostate Tumor Cells and Its Clinical and Biological Significance in Prostate Cancer Progression. J Cancer Sci Ther. 2012 Apr 26;4(4):94-101. doi: 10.4172/1948-5956.1000119. PMID:23264855 doi:http://dx.doi.org/10.4172/1948-5956.1000119
- ↑ Tomlins SA, Rhodes DR, Perner S, Dhanasekaran SM, Mehra R, Sun XW, Varambally S, Cao X, Tchinda J, Kuefer R, Lee C, Montie JE, Shah RB, Pienta KJ, Rubin MA, Chinnaiyan AM. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science. 2005 Oct 28;310(5748):644-8. doi: 10.1126/science.1117679. PMID:16254181 doi:http://dx.doi.org/10.1126/science.1117679
- ↑ Carrere S, Verger A, Flourens A, Stehelin D, Duterque-Coquillaud M. Erg proteins, transcription factors of the Ets family, form homo, heterodimers and ternary complexes via two distinct domains. Oncogene. 1998 Jun 25;16(25):3261-8. doi: 10.1038/sj.onc.1201868. PMID:9681824 doi:http://dx.doi.org/10.1038/sj.onc.1201868
- ↑ Yu J, Yu J, Mani RS, Cao Q, Brenner CJ, Cao X, Wang X, Wu L, Li J, Hu M, Gong Y, Cheng H, Laxman B, Vellaichamy A, Shankar S, Li Y, Dhanasekaran SM, Morey R, Barrette T, Lonigro RJ, Tomlins SA, Varambally S, Qin ZS, Chinnaiyan AM. An integrated network of androgen receptor, polycomb, and TMPRSS2-ERG gene fusions in prostate cancer progression. Cancer Cell. 2010 May 18;17(5):443-54. doi: 10.1016/j.ccr.2010.03.018. PMID:20478527 doi:http://dx.doi.org/10.1016/j.ccr.2010.03.018
- ↑ Farooqi AA, Hou MF, Chen CC, Wang CL, Chang HW. Androgen receptor and gene network: Micromechanics reassemble the signaling machinery of TMPRSS2-ERG positive prostate cancer cells. Cancer Cell Int. 2014 Apr 17;14:34. doi: 10.1186/1475-2867-14-34. eCollection, 2014. PMID:24739220 doi:http://dx.doi.org/10.1186/1475-2867-14-34
- ↑ Thunders M, Delahunt B. Gene of the month: TMPRSS2 (transmembrane serine protease 2). J Clin Pathol. 2020 Dec;73(12):773-776. doi: 10.1136/jclinpath-2020-206987. Epub , 2020 Sep 1. PMID:32873700 doi:http://dx.doi.org/10.1136/jclinpath-2020-206987
- ↑ Hoffmann M, Schroeder S, Kleine-Weber H, Muller MA, Drosten C, Pohlmann S. Nafamostat Mesylate Blocks Activation of SARS-CoV-2: New Treatment Option for COVID-19. Antimicrob Agents Chemother. 2020 May 21;64(6). pii: AAC.00754-20. doi:, 10.1128/AAC.00754-20. Print 2020 May 21. PMID:32312781 doi:http://dx.doi.org/10.1128/AAC.00754-20
- ↑ Thunders M, Delahunt B. Gene of the month: TMPRSS2 (transmembrane serine protease 2). J Clin Pathol. 2020 Dec;73(12):773-776. doi: 10.1136/jclinpath-2020-206987. Epub , 2020 Sep 1. PMID:32873700 doi:http://dx.doi.org/10.1136/jclinpath-2020-206987
- ↑ Shen LW, Mao HJ, Wu YL, Tanaka Y, Zhang W. TMPRSS2: A potential target for treatment of influenza virus and coronavirus infections. Biochimie. 2017 Nov;142:1-10. doi: 10.1016/j.biochi.2017.07.016. Epub 2017 Aug 1. PMID:28778717 doi:http://dx.doi.org/10.1016/j.biochi.2017.07.016
- ↑ Amraei R, Rahimi N. COVID-19, Renin-Angiotensin System and Endothelial Dysfunction. Cells. 2020 Jul 9;9(7). pii: cells9071652. doi: 10.3390/cells9071652. PMID:32660065 doi:http://dx.doi.org/10.3390/cells9071652
- ↑ Shrimp JH, Kales SC, Sanderson PE, Simeonov A, Shen M, Hall MD. An Enzymatic TMPRSS2 Assay for Assessment of Clinical Candidates and Discovery of Inhibitors as Potential Treatment of COVID-19. bioRxiv. 2020 Aug 6. doi: 10.1101/2020.06.23.167544. PMID:32596694 doi:http://dx.doi.org/10.1101/2020.06.23.167544
- ↑ Hitomi Y, Ikari N, Fujii S. Inhibitory effect of a new synthetic protease inhibitor (FUT-175) on the coagulation system. Haemostasis. 1985;15(3):164-8. doi: 10.1159/000215139. PMID:3161808 doi:http://dx.doi.org/10.1159/000215139
- ↑ doi: https://dx.doi.org/10.1101/2021.06.23.449282
- ↑ doi: https://dx.doi.org/10.1101/2021.06.23.449282
- ↑ Hoffmann M, Schroeder S, Kleine-Weber H, Muller MA, Drosten C, Pohlmann S. Nafamostat Mesylate Blocks Activation of SARS-CoV-2: New Treatment Option for COVID-19. Antimicrob Agents Chemother. 2020 May 21;64(6). pii: AAC.00754-20. doi:, 10.1128/AAC.00754-20. Print 2020 May 21. PMID:32312781 doi:http://dx.doi.org/10.1128/AAC.00754-20
- ↑ Hoffmann M, Schroeder S, Kleine-Weber H, Muller MA, Drosten C, Pohlmann S. Nafamostat Mesylate Blocks Activation of SARS-CoV-2: New Treatment Option for COVID-19. Antimicrob Agents Chemother. 2020 May 21;64(6). pii: AAC.00754-20. doi:, 10.1128/AAC.00754-20. Print 2020 May 21. PMID:32312781 doi:http://dx.doi.org/10.1128/AAC.00754-20
- ↑ Shrimp JH, Kales SC, Sanderson PE, Simeonov A, Shen M, Hall MD. An Enzymatic TMPRSS2 Assay for Assessment of Clinical Candidates and Discovery of Inhibitors as Potential Treatment of COVID-19. bioRxiv. 2020 Aug 6. doi: 10.1101/2020.06.23.167544. PMID:32596694 doi:http://dx.doi.org/10.1101/2020.06.23.167544
- ↑ doi: https://dx.doi.org/10.1101/2021.06.23.449282
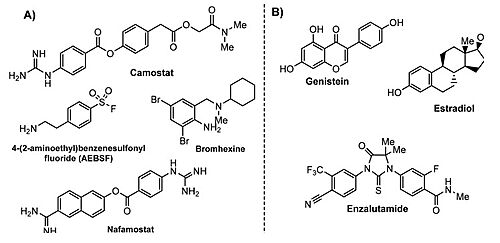
- ↑ Maggio R, Corsini GU. Repurposing the mucolytic cough suppressant and TMPRSS2 protease inhibitor bromhexine for the prevention and management of SARS-CoV-2 infection. Pharmacol Res. 2020 Jul;157:104837. doi: 10.1016/j.phrs.2020.104837. Epub 2020, Apr 22. PMID:32334052 doi:http://dx.doi.org/10.1016/j.phrs.2020.104837
- ↑ doi: https://dx.doi.org/10.1101/2021.06.23.449282
- ↑ doi: https://dx.doi.org/10.20944/preprints202003.0360.v2
- ↑ Gil C, Ginex T, Maestro I, Nozal V, Barrado-Gil L, Cuesta-Geijo MA, Urquiza J, Ramirez D, Alonso C, Campillo NE, Martinez A. COVID-19: Drug Targets and Potential Treatments. J Med Chem. 2020 Nov 12;63(21):12359-12386. doi: 10.1021/acs.jmedchem.0c00606., Epub 2020 Jun 26. PMID:32511912 doi:http://dx.doi.org/10.1021/acs.jmedchem.0c00606
Proteopedia Page Contributors and Editors (what is this?)
Paula R. Mallavibarrena, Ines Muniesa-Martinez, Laura Aleixos Juan, Michal Harel, Jaime Prilusky