Function
The Bam (β-Barrel Assembly Machinery) drives the assembly of β-barrel proteins into the outer membrane of gram-negative bacteria.[1]
Structural highlights
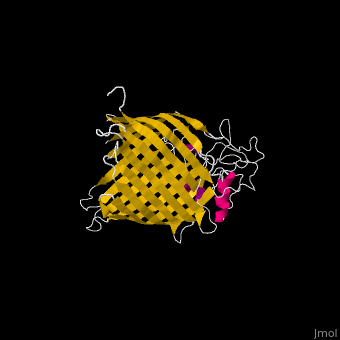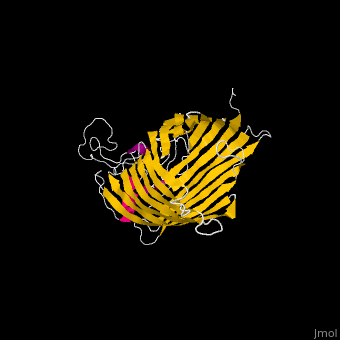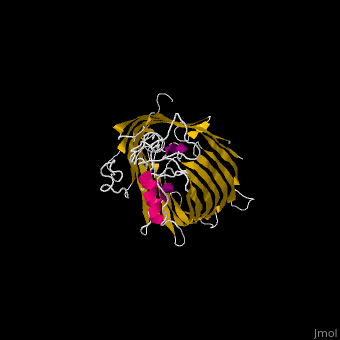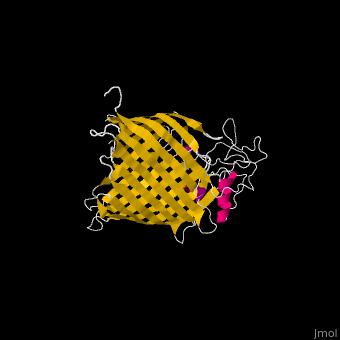
The complex is composed of five subunits: BamA, BamB, BamC, BamD and BamE. Outer membrane β-barrel proteins assembly is dependent on Bam in various organisms. BamB,C,D,E bind to the N-terminal of BamA.
- BamA contains five structurally homologous POTRA (POlypeptide TRAnslocation associated) domains. The POTRA domain has a β-α-α-β-β conformation. BamA barrel and at least a subset of its POTRAs are essential for viability. BamA was found also in mitochondria and chloroplasts.
- BamD is composed of multiple tetratricopeptide (TPR) repeats packed into a superhelical structure. TPR is a motif containing 2 antiparallel α-helices. TPRs are found in scaffold multiprotein complexes and are involved in protein-protein interactions.
3D Structures of Bam complex
Bam complex 3D structures