| Structural highlights
Function
RACK1_HUMAN Involved in the recruitment, assembly and/or regulation of a variety of signaling molecules. Interacts with a wide variety of proteins and plays a role in many cellular processes. Component of the 40S ribosomal subunit involved in translational repression. Binds to and stabilizes activated protein kinase C (PKC), increasing PKC-mediated phosphorylation. May recruit activated PKC to the ribosome, leading to phosphorylation of EIF6. Inhibits the activity of SRC kinases including SRC, LCK and YES1. Inhibits cell growth by prolonging the G0/G1 phase of the cell cycle. Enhances phosphorylation of BMAL1 by PRKCA and inhibits transcriptional activity of the BMAL1-CLOCK heterodimer. Facilitates ligand-independent nuclear translocation of AR following PKC activation, represses AR transactivation activity and is required for phosphorylation of AR by SRC. Modulates IGF1R-dependent integrin signaling and promotes cell spreading and contact with the extracellular matrix. Involved in PKC-dependent translocation of ADAM12 to the cell membrane. Promotes the ubiquitination and proteasome-mediated degradation of proteins such as CLEC1B and HIF1A. Required for VANGL2 membrane localization, inhibits Wnt signaling, and regulates cellular polarization and oriented cell division during gastrulation. Required for PTK2/FAK1 phosphorylation and dephosphorylation. Regulates internalization of the muscarinic receptor CHRM2. Promotes apoptosis by increasing oligomerization of BAX and disrupting the interaction of BAX with the anti-apoptotic factor BCL2L. Inhibits TRPM6 channel activity. Regulates cell surface expression of some GPCRs such as TBXA2R. Plays a role in regulation of FLT1-mediated cell migration. Involved in the transport of ABCB4 from the Golgi to the apical bile canalicular membrane (PubMed:19674157). Binds to Y.pseudotuberculosis yopK which leads to inhibition of phagocytosis and survival of bacteria following infection of host cells. Enhances phosphorylation of HIV-1 Nef by PKCs. Promotes migration of breast carcinoma cells by binding to and activating RHOA.[1] [2] [3] [4] [5] [6] [7] [8] [9] [10] [11] [12] [13] [14] [15] [16] [17] [18] [19] [20]
Publication Abstract from PubMed
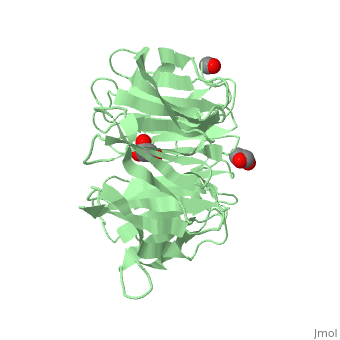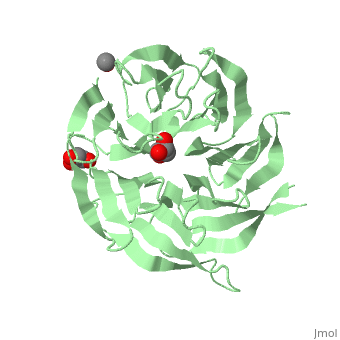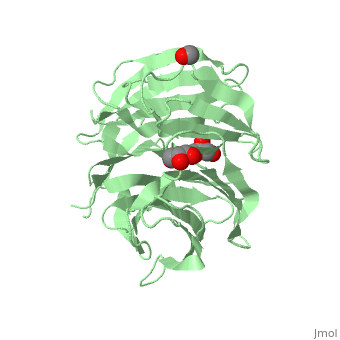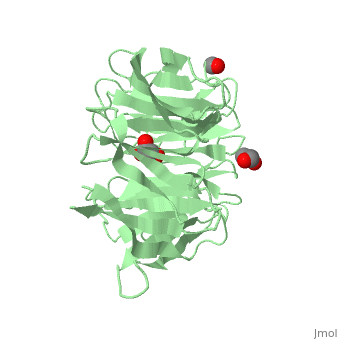
The crystal structure of human receptor for activated C-kinase 1 (hRack1) protein is reported at 2.45 A resolution. The crystals belongs to space group P4(1)2(1)2, with three molecules per asymmetric unit. The hRack1 structure features a sevenfold beta-propeller, with each blade housing a sequence motif that contains a strictly conserved Trp, the indole group of which is embedded between adjacent blades. In blades 1-5 the imidazole group of a His residue is wedged between the side chains of a Ser residue and an Asp residue through two hydrogen bonds. The hRack1 crystal structure forms a starting basis for understanding the remarkable scaffolding properties of this protein.
Structure of human Rack1 protein at a resolution of 2.45 A.,Ruiz Carrillo D, Chandrasekaran R, Nilsson M, Cornvik T, Liew CW, Tan SM, Lescar J Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012 Aug 1;68(Pt 8):867-72., Epub 2012 Jul 26. PMID:22869111[21]
From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.
See Also
References
- ↑ Gallina A, Rossi F, Milanesi G. Rack1 binds HIV-1 Nef and can act as a Nef-protein kinase C adaptor. Virology. 2001 Apr 25;283(1):7-18. PMID:11312657 doi:10.1006/viro.2001.0855
- ↑ Hermanto U, Zong CS, Li W, Wang LH. RACK1, an insulin-like growth factor I (IGF-I) receptor-interacting protein, modulates IGF-I-dependent integrin signaling and promotes cell spreading and contact with extracellular matrix. Mol Cell Biol. 2002 Apr;22(7):2345-65. PMID:11884618
- ↑ Cox EA, Bennin D, Doan AT, O'Toole T, Huttenlocher A. RACK1 regulates integrin-mediated adhesion, protrusion, and chemotactic cell migration via its Src-binding site. Mol Biol Cell. 2003 Feb;14(2):658-69. PMID:12589061 doi:10.1091/mbc.E02-03-0142
- ↑ Rigas AC, Ozanne DM, Neal DE, Robson CN. The scaffolding protein RACK1 interacts with androgen receptor and promotes cross-talk through a protein kinase C signaling pathway. J Biol Chem. 2003 Nov 14;278(46):46087-93. Epub 2003 Sep 4. PMID:12958311 doi:http://dx.doi.org/10.1074/jbc.M306219200
- ↑ Kraus S, Gioeli D, Vomastek T, Gordon V, Weber MJ. Receptor for activated C kinase 1 (RACK1) and Src regulate the tyrosine phosphorylation and function of the androgen receptor. Cancer Res. 2006 Nov 15;66(22):11047-54. PMID:17108144 doi:10.1158/0008-5472.CAN-06-0596
- ↑ Liu YV, Baek JH, Zhang H, Diez R, Cole RN, Semenza GL. RACK1 competes with HSP90 for binding to HIF-1alpha and is required for O(2)-independent and HSP90 inhibitor-induced degradation of HIF-1alpha. Mol Cell. 2007 Jan 26;25(2):207-17. PMID:17244529 doi:10.1016/j.molcel.2007.01.001
- ↑ Chuang NN, Huang CC. Interaction of integrin beta1 with cytokeratin 1 in neuroblastoma NMB7 cells. Biochem Soc Trans. 2007 Nov;35(Pt 5):1292-4. PMID:17956333 doi:10.1042/BST0351292
- ↑ Parent A, Laroche G, Hamelin E, Parent JL. RACK1 regulates the cell surface expression of the G protein-coupled receptor for thromboxane A(2). Traffic. 2008 Mar;9(3):394-407. Epub 2007 Dec 14. PMID:18088317 doi:10.1111/j.1600-0854.2007.00692.x
- ↑ Cao G, Thebault S, van der Wijst J, van der Kemp A, Lasonder E, Bindels RJ, Hoenderop JG. RACK1 inhibits TRPM6 activity via phosphorylation of the fused alpha-kinase domain. Curr Biol. 2008 Feb 12;18(3):168-76. doi: 10.1016/j.cub.2007.12.058. PMID:18258429 doi:10.1016/j.cub.2007.12.058
- ↑ Bourd-Boittin K, Le Pabic H, Bonnier D, L'Helgoualc'h A, Theret N. RACK1, a new ADAM12 interacting protein. Contribution to liver fibrogenesis. J Biol Chem. 2008 Sep 19;283(38):26000-9. doi: 10.1074/jbc.M709829200. Epub 2008 , Jul 11. PMID:18621736 doi:10.1074/jbc.M709829200
- ↑ Kiely PA, Baillie GS, Barrett R, Buckley DA, Adams DR, Houslay MD, O'Connor R. Phosphorylation of RACK1 on tyrosine 52 by c-Abl is required for insulin-like growth factor I-mediated regulation of focal adhesion kinase. J Biol Chem. 2009 Jul 24;284(30):20263-74. doi: 10.1074/jbc.M109.017640. Epub, 2009 May 7. PMID:19423701 doi:10.1074/jbc.M109.017640
- ↑ Ikebuchi Y, Takada T, Ito K, Yoshikado T, Anzai N, Kanai Y, Suzuki H. Receptor for activated C-kinase 1 regulates the cellular localization and function of ABCB4. Hepatol Res. 2009 Nov;39(11):1091-107. doi: 10.1111/j.1872-034X.2009.00544.x., Epub 2009 Aug 6. PMID:19674157 doi:http://dx.doi.org/10.1111/j.1872-034X.2009.00544.x
- ↑ Ruan Y, Guo L, Qiao Y, Hong Y, Zhou L, Sun L, Wang L, Zhu H, Wang L, Yun X, Xie J, Gu J. RACK1 associates with CLEC-2 and promotes its ubiquitin-proteasome degradation. Biochem Biophys Res Commun. 2009 Dec 11;390(2):217-22. doi:, 10.1016/j.bbrc.2009.09.087. Epub 2009 Sep 26. PMID:19785988 doi:10.1016/j.bbrc.2009.09.087
- ↑ Cao XX, Xu JD, Xu JW, Liu XL, Cheng YY, Li QQ, Xu ZD, Liu XP. RACK1 promotes breast carcinoma migration/metastasis via activation of the RhoA/Rho kinase pathway. Breast Cancer Res Treat. 2011 Apr;126(3):555-63. doi: 10.1007/s10549-010-0955-3. , Epub 2010 May 25. PMID:20499158 doi:10.1007/s10549-010-0955-3
- ↑ Wu Y, Wang Y, Sun Y, Zhang L, Wang D, Ren F, Chang D, Chang Z, Jia B. RACK1 promotes Bax oligomerization and dissociates the interaction of Bax and Bcl-XL. Cell Signal. 2010 Oct;22(10):1495-501. doi: 10.1016/j.cellsig.2010.05.018. Epub, 2010 Jun 10. PMID:20541605 doi:10.1016/j.cellsig.2010.05.018
- ↑ Schaffler K, Schulz K, Hirmer A, Wiesner J, Grimm M, Sickmann A, Fischer U. A stimulatory role for the La-related protein 4B in translation. RNA. 2010 Aug;16(8):1488-99. doi: 10.1261/rna.2146910. Epub 2010 Jun 23. PMID:20573744 doi:10.1261/rna.2146910
- ↑ Reiner CL, McCullar JS, Kow RL, Le JH, Goodlett DR, Nathanson NM. RACK1 associates with muscarinic receptors and regulates M(2) receptor trafficking. PLoS One. 2010 Oct 20;5(10):e13517. doi: 10.1371/journal.pone.0013517. PMID:20976005 doi:10.1371/journal.pone.0013517
- ↑ Wang F, Yamauchi M, Muramatsu M, Osawa T, Tsuchida R, Shibuya M. RACK1 regulates VEGF/Flt1-mediated cell migration via activation of a PI3K/Akt pathway. J Biol Chem. 2011 Mar 18;286(11):9097-106. doi: 10.1074/jbc.M110.165605. Epub, 2011 Jan 6. PMID:21212275 doi:10.1074/jbc.M110.165605
- ↑ Thorslund SE, Edgren T, Pettersson J, Nordfelth R, Sellin ME, Ivanova E, Francis MS, Isaksson EL, Wolf-Watz H, Fallman M. The RACK1 signaling scaffold protein selectively interacts with Yersinia pseudotuberculosis virulence function. PLoS One. 2011 Feb 10;6(2):e16784. doi: 10.1371/journal.pone.0016784. PMID:21347310 doi:10.1371/journal.pone.0016784
- ↑ Chang BY, Conroy KB, Machleder EM, Cartwright CA. RACK1, a receptor for activated C kinase and a homolog of the beta subunit of G proteins, inhibits activity of src tyrosine kinases and growth of NIH 3T3 cells. Mol Cell Biol. 1998 Jun;18(6):3245-56. PMID:9584165
- ↑ Ruiz Carrillo D, Chandrasekaran R, Nilsson M, Cornvik T, Liew CW, Tan SM, Lescar J. Structure of human Rack1 protein at a resolution of 2.45 A. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2012 Aug 1;68(Pt 8):867-72., Epub 2012 Jul 26. PMID:22869111 doi:10.1107/S1744309112027480
|