Function
Aminopeptidases (AP) (EC 3) are metal - mostly Zn-dependent enzymes involved in the digestion of proteins. Cytosol AP (Cyt-AP), deblocking AP (DAP) and AP N (APN) remove N-terminal amino acids. The AP are classified by the amino acid which they hydrolyze. Other types of AP are:
Aminopeptidases catalyze a release of an N-terminal amino acid from a peptide, amide, or arylamide. Beta-peptidyl AP (BapA) cleaves N-terminal β-homoamino acid from peptides of length 2 to 6.[1]
Aminopeptidase from Aeromonas proteolytica[2]
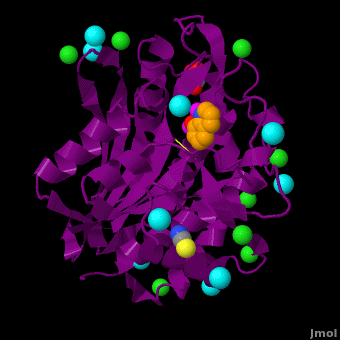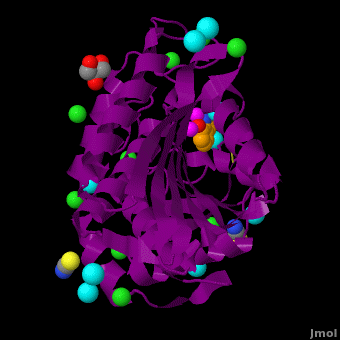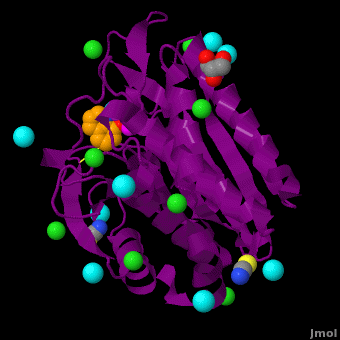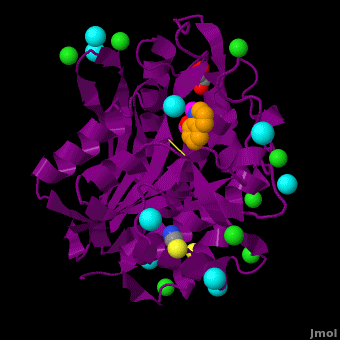
The selective inhibition of an , a hydrolase, by derivatives is reported. Based on our findings about 8-HQ-based Zn2+ fluorophores, it was hypothesized that 8-HQ derivatives have the potential to function as specific inhibitors of Zn2+ enzymes, especially dinuclear Zn2+ hydrolases. Inhibitory assays of 8-HQ derivatives against AAP disclosed that the 8-HQ and 5-substituted 8-HQ′s are competitive inhibitors for AAP with inhibition constants (Ki) of 0.16—29 μM at pH 8.0. (1.3 Å resolution) as well as fluorescence titrations of these drugs with AAP confirmed that , in which the in the active site of AAP and the (PDB code: 3vh9). of free AAP (colored green) containing Zn2+-bound water molecule (H2O or OH-; red sphere) (1rtq) bridging two Zn2+ and AAP–8-HQ complex (darkmagenta, 3vh9). Two Zn2+ are depicted as magenta spheres.
S. griseus aminopeptidase
S. griseus aminopeptidase (SGAP) cleaves the N-terminal amino acid from a peptide or protein, and is specific for larger hydrophobic acids, especially leucine. No cleavage occurs if the next residue is proline.
The of the enzyme contains two Zn2+ ions with His85 and Asp160 as ligands for one ion, and Glu132 and His247 as ligands for the second ion. Asp97 is a common ligand to both ions. What appears to be a phosphate anion is bound to both zinc atoms, replacing the water molecule/hydroxide ion normally found in this class of enzyme. See details of SGAP in Streptomyces griseus Aminopeptidase (SGAP).
3D Structures of Aminopeptidase
Aminopeptidase 3D structures